Research Interests
I feel strongly attracted by the research in the fields of natural sciences and most specifically in physics, chemistry and astronomy. Thus, astrophysical and astrochemical questions have been the main driver of my research including laboratory work on the production and spectroscopic characterization of astrophysical relevant molecules as well as their astrophysical detection using radio(mm)-telescopes .
Projects

(picture credit NASA/JPL)
High-resolution Spectroscopy using Chirped-Pulse Fourier Transform Technique
We perform dedicated experiments in the 100 GHz region on transient, mostly refractory materials (C, Si, metals, ) bearing molecules.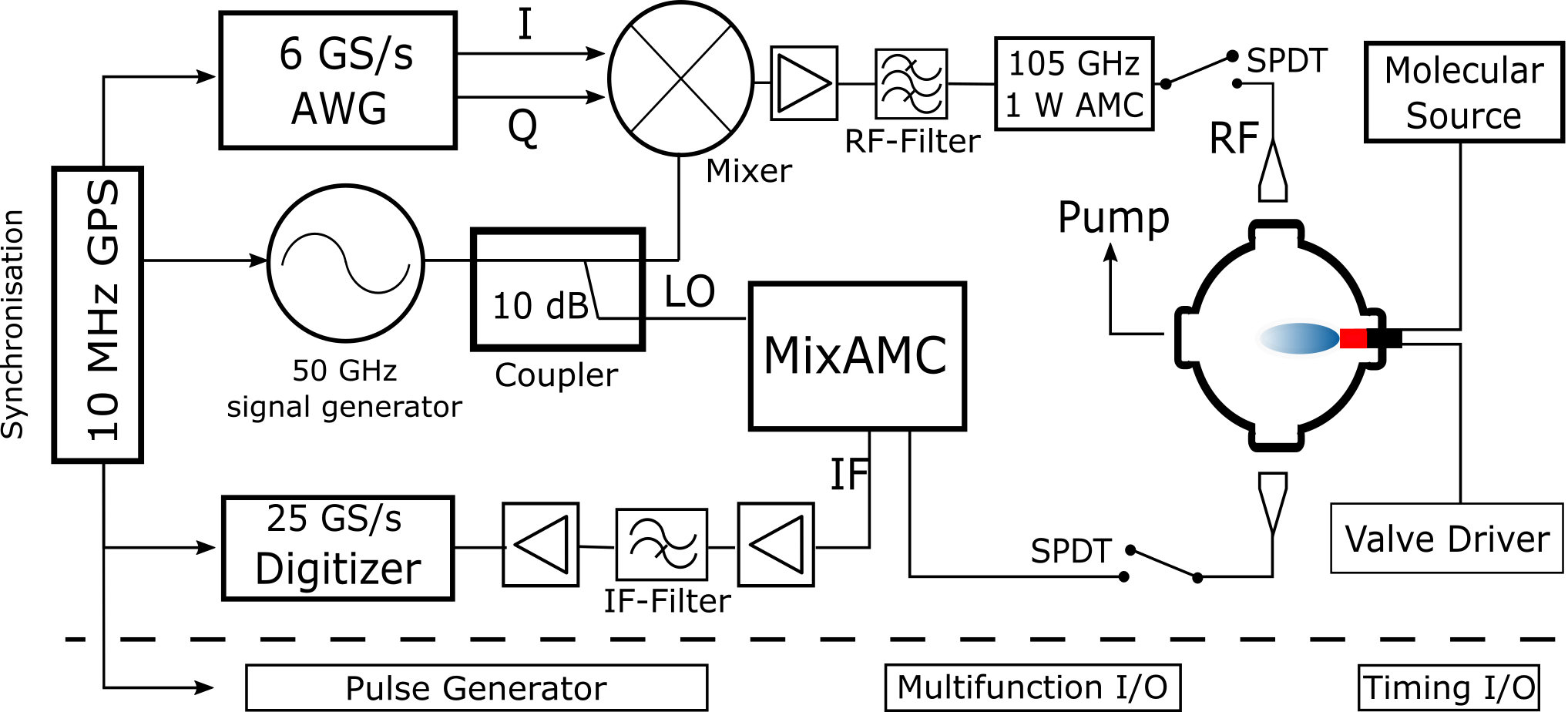
(CP-FTMM experimenatal setup, 2018, by Fuchs)
Infrared Cavity Ringdown Spectroscopy
There is a surprisingly rich chemistry to be found in different regions in outer space. For example, in the vicinity of dying stars atoms combine to molecules and serve as seed material for dust formation (e.g. silicon carbides). Unlike on earth, interstellar molecules are very often non-saturated compounds that are highly reactive. These species, which are difficult to produce in terrestrial laboratories, are valuable probes to determine the local physical conditions in cosmic environments. Many of the molecules have characteristic spectra at mid-IR wavelengths. These spectra cannot be calculated by purely theoretical means to the necessary level of precision for astrophysical detections. In this work we perform high-resolution and extremely sensitive measurements on astrophysically relevant molecules. Powerful lasers, like OPO systems for the region 2-4.7 mu are utilized. These laser systems are an integral part of the new highly sensitive cavity-ringdown (CRD) spectrometer. The IR-CRD spectrometer is combined with special plasma molecule sources (laser ablation/electr. discharge) to produce in-situ high molecule abundances.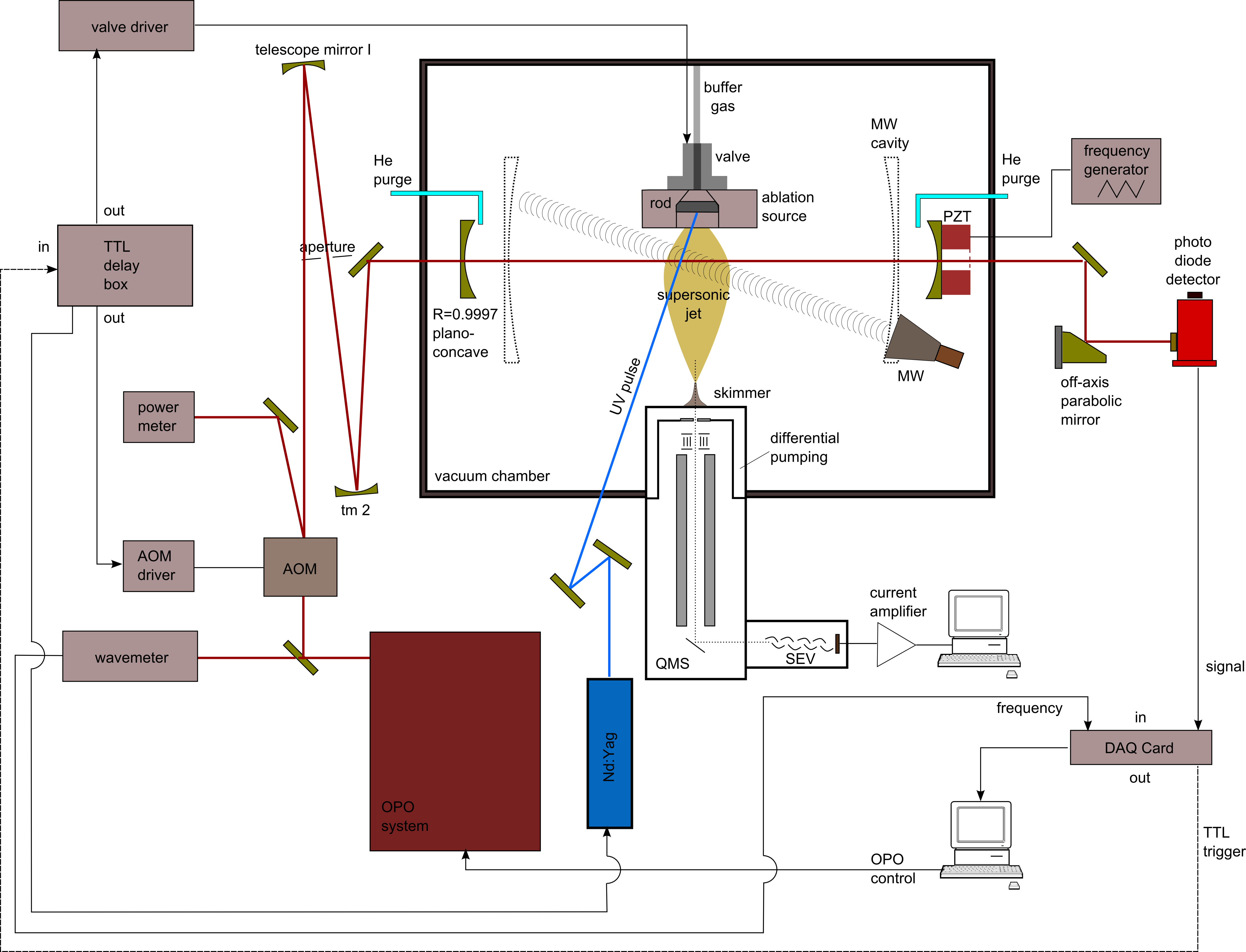
(IR-CRD experimenatal setup, 2017, by Fuchs)
Surface reactions on Interstellar Ice analogs
The formation of stars and planets is one of the most exciting fields of research of modern days astrophysics. Properties of the early evolutionary stages, in particular the early chemical development is of paramount interest for the nowadays composition of the planets and particularly of the Earth. For decades chemical complexity in space has been explained by ion-neutral reactions in the gas-phase. Exciting new evidence in the field of observational astronomy and laboratory astrophysics has changed this picture significantly in recent years. It is now clear that not only H2 but many other and more complex molecules have to be formed on cold interstellar grain surfaces. The here presented research is aimed to simulate the formation of complex molecules of astrophysical interest on cryogenic surfaces like interstellar ice analogues (solid H2O, CO2 and CO) under controlled laboratory conditions. Atom-atom and molecule-atom reactions are examined since these form the key to complex and pre-biotic molecule formation in a dense interstellar environment. At the Leiden Observatory the Sackler Laboratory was founded in 1975 by M. Greenberg to specifically conduct experiments on interstellar ice analogs. Today this Laboratory is headed by E.F. van Dishoeck and H. Linnartz. In the two years of my post-doc time in Leiden I designed and constructed the new ultra-high vacuum experiment, SURFRESIDE II. Here, cryogenic solids ('ices') are grown and bombard with H atoms, using the most efficient and best characterized H atom source yet available. Reaction products and pathways, as well as possible reaction barriers, are examined using well established sensitive surface analysing techniques. Formation schemes of molecules like methanol, formaldehyde and ethanol are topics of a current research project. The experimental results provide important molecular data needed as input for chemical and astrochemical modelling. Many, if not most, of the important grain-surface reactions in space have not yet been classified thus preventing further progress in that field. Future results will have a significant impact on the analysis of astronomical observations from upcoming facilities like ALMA, Herschel and eSMA.

(Surfreside II, 2006, by Fuchs)
The experimental apparatus: SURFRESIDE II
This experiment is designed to investigate surface reactions of atoms and radicals on cryogenic surfaces. SURFRESIDE II is equiped with a quadrupole mass spectrometer (QMS) and an IR-Fourier transform spectrometer (IR-FTS). Hydrogen atoms are produced by a thermal cracking source and are subsequently cooled to 100 K by wall collisions. First studies have also been done with a microwave (MW) discharge source and in the future it will be possible to produce atoms like O and N or other radicals. As a target for the atom/radicals a thin gold surface, which can be cooled down to 10 K, is placed in the middle of an ultrahigh vacuum (UHV) chamber. Volatile gas molecules can condense on this cold surface and form amorpheous or crystalline solids (matrix, ice). Any reaction of the atoms and radicals on or with the ice can be observed using reflection absorption IR spectroscopy (RAIRS, IRRAS). Evaporated species can be monitored by the QMS and give further inside on the ongoing surface reactions, i.e. temperature programmed desorption (TPD) experiments.
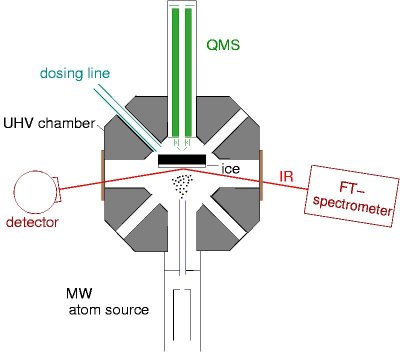 | SURFRESIDE II, experimental setup (January 2007) by G.W.Fuchs. The QMS (quadrupole mass spectrometer) is used for TPD measuremends. The FT-IR spectrometer is set up in an absorption-reflection-absorption mode. The atom source can be either a thermal cracking solurce or an MW discharge source (shown). |
Surface reaction experiments
The reaction of CO + H into H2CO and CH3OH has been investigated using the RAIRS and TPD technique. The produced species were identified according to their IR fingerprint and mass spectra. Furthermore, the species abundances as a function of atom bombardment time has been determined. Different H atom fluxes, morphologies, ice thicknesses and deposition methods have been tested. The experimental results were analyzed using rate equations. The formation of formaldehyde and methanol via CO + H into H2CO and CH3OH at low temperatures can be confirmed and their reaction rates have been determined. For CO ices thicker than 10 monolayers, the reaction rate of H2CO as well as that of CH3OH does not change as a function of ice thickness. The functional surface temperature dependence of the reactions shows a maximum reaction rate for both, formaldehyde and methanol, around 13 - 15 K. Different H fluxes were tested for pure CO ices and a nearly constant CO to H2CO reaction rate was found. Co-deposition of CO and H shows similar H2CO and CH3OH production rates as a 'first deposit then bombard' approach. All this information can be used to model real interstellar ices and their chemical behaviour. The results can be directly compared to astronomical observations.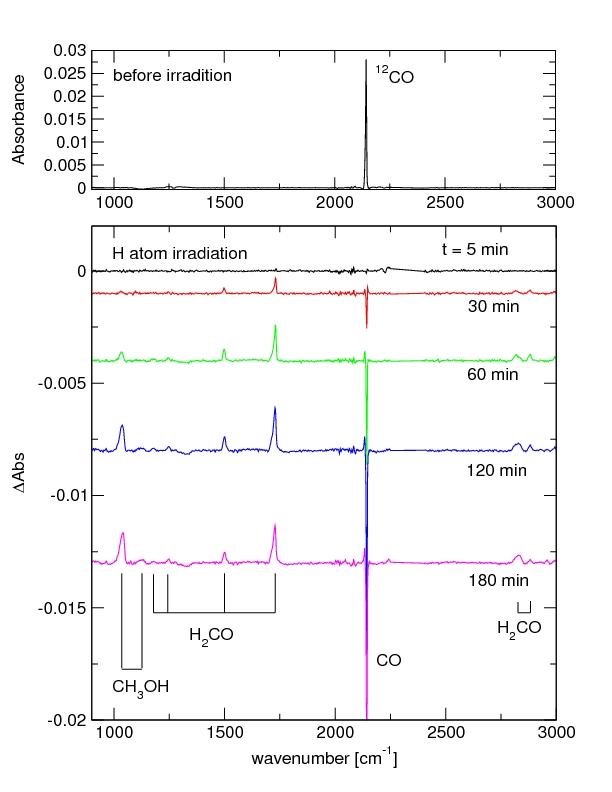
(First experimental results from SURFRESIDE II. IR spectra of the hydrogenation of 70 monolayer of pure CO at 15 K surface temperature. Top: CO after deposition. Bottom: Growth of formaldehyde and methanol signals during H irradiation, Fuchs et al. 2007)
Acknowledgements
W. Schutte initiated the SURFRESIDE experiment and S.Schlemmer and H. Fraser constructed SURFRESIDE I. S.Schlemmer was particularly helpful with his suggestions and experience for the design of SURFRESIDE II. J. Disselhorst, S. Bisschop, S. Ioppolo and H. Linnartz were very helpful during the build-up of SURFRESIDE II. In addition many experiments have been performed using the CRYOPAD experimental setup, e.g. UV photodesorption of CO by K. Oberg and O2 thermal desorption by K. Acharyya. Special thanks to E.F. van Dishoeck who gave me the opportunity to work in Leiden for more then 2 years.
Astronomical Observations in the submm region
Motivated by the laboratory work in the MW wavelength region I started two astronomical projects. The first one was the search for the linear C2N radical towards the circumstellar envelope IRC+10216. Eight frequency bands of the rotational transitions J = 7/2 - 5/2 up to 13/2 - 11/2 and 17/2 - 15/2 up to 23/2 - 21/2 of the C2N molecule including their lambda-doubling components have been probed, using the IRAM 30 m telescope at Pico Veleta, Spain. However, there is no evidence for the existence of the C2N radical towards IRC+10216. Instead, a new upper limit N = 3.8*10(-12) cm-2 for the column density of C2N was estimated. Additionally, at least ten yet unidentified lines have been found.
(The Iram 30m telescope, 2003)
The second project was an extensive search for the complex molecule trans-ethyl methyl ether towards several hot core regions. Using the IRAM 30 m telescope and the SEST 15 m a search was performed at several frequencies where trans-ethyl methyl ether has strong transitions, as well as lines which are particularly sensitive to the physical conditions in which the molecule can be found. The target sources were G34.26, NGC 6334(I), Orion KL, and W51e2 which have been proven to have a rich chemistry of complex molecules. Our observations could not confirm the tentative Orion KL detection previously made by Charnley et al. (2001) within their stated column density limits, but the existence of the trans-ethyl methyl ether towards W51e2 with a column density of 2*10(14) cm-2 could be confirmed. The dimethyl ether/methanol ratio of 0.6 as well as the newly found ethyl methyl ether/ethanol ratio of 0.13 indicate relative high abundances of ethers toward W51e2. Furthermore, the observation of ethyl methyl ether also confirms the importance of ethanol as a grain mantle constituent. New upper limits for the column densities of the molecule of around 8*10(13) cm-2 were found toward Orion KL, G34.26, NGC6334(I) and the estimated column density towards SgrB2(N) is of the same order.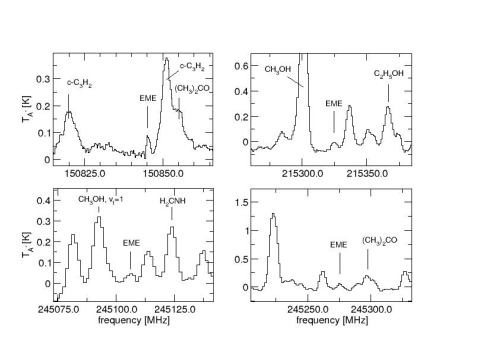
Spectra recorded towards W51e2. The methyl-ethyl ether transitions 20(0,20)-19(1,19) at 150 845.5 MHz, 19(5,14)-19(4,15) at 215 325.4 MHz, 16(3,14)-15(2,13) at 245 106.4 MHz, and 17(3,15)-16(2,14) at 245 190.7 MHz are indicated.
Acknowledgements
I am deeply indepted to F. Wyrowski, P. Schilke and my wife U. Fuchs who helped me realizing these astronomical projects.
MW spectroscopy of small carbon chains
 |
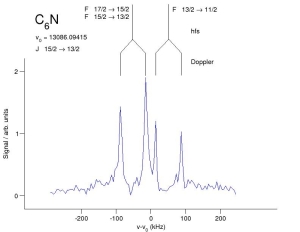 |
The Harvard discharge nozzle during operation. | Spectra of C6N at 13 GHz. |
At Harvard I worked in the spectroscopy group of P. Thaddeus and M. McCarthy. During that time we detected two new carbon chain radicals C4N and C6N terminated with a nitrile group. The experiments were performed using a supersonic molecular beam produced by a discharge nozzle (see picture) and a Fourier transform microwave spectrometer (see figure). Both C4N and C6N are linear chains with 2Pi electronic ground states, and both radicals have resolvable hyperfine structure and lambda-type doubling in their lowest rotational levels. At least four transitions in the lowest-energy fine structure component (2P1/2) were measured between 7 and 22 GHz for both molecules, and at most nine spectroscopic constants were required to reproduce the measured spectra to a few parts in 10(7). In addition, at least three hyperfine-split rotational transitions of the singly-substituted isotopic species of C3N have been observed. Precise sets of spectroscopic constants were also determined for the C3N isotopomers by combining the centimeter-wave measurements with previous millimeter-wave data. Although the strongest lines of C6N are more than five times less intense than those of C5N, owing to large differences in the ground state dipole moments, the newly found chains C4N and C6N are more abundant than C5N. So far, searches for C7N have been unsuccessful. The absence of lines at the predicted frequencies is indicative that the ground state of C7N may be 2P, for which the dipole moment is calculated to be small.
The Fourier Transform Microwave spectrometer at Harvard, 2001.
Acknowledgements
Many tanks to E.S. Palmer, M.E. Sanz, J Dudek and the Gottliebs who always were helpful and never afraid of an interesting discussion.
IR spectroscopy of Linear Carbon clusters
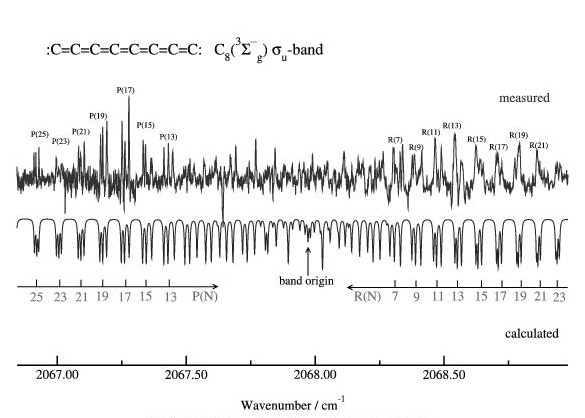
Mid-IR Spectrum of linear C8, (Neubauer-Guenther et al. 2002)
The Cologene Carbon Cluster IR spectrometer was designed to investigate the precise structures of small (C10 - C60) carbon clusters. Important questions such as how Bucky balls (C60) can form and which possible intermediate structures exist can be addressed. Although calculations favour the existance of ring- or cave-like structures for larger carbon cluster (> C10) so far only linear species have been detected. During my time in Cologne C8, C9, C10, and C13 have been (re-)detected. The experiments were performed as follows. A graphite laser ablation source produces pure carbon clusters of all sizes in a cold supersonic jet of an inert gas (e.g He). The IR absorption signal is recorded by detecting the light of a tunable IR diode which intersects the cluster jet 20 - 40 times in a Herriott-type multipass optical system. The signals from three HgCdTe photoconducting detectors, the sample, the reference gas (OCS), and the etalon signal for frequency calibration were collected simultaneously and stored by a computer. Signal averaging over 30 - 40 pulses of the ablation source and the use of a bandpass filter have increased the signal-to-noise ratio significantly. The spectral resolution was better than 10(+4) cm-1 and the absolute frequency accuracy better than 10(+3) cm-1.
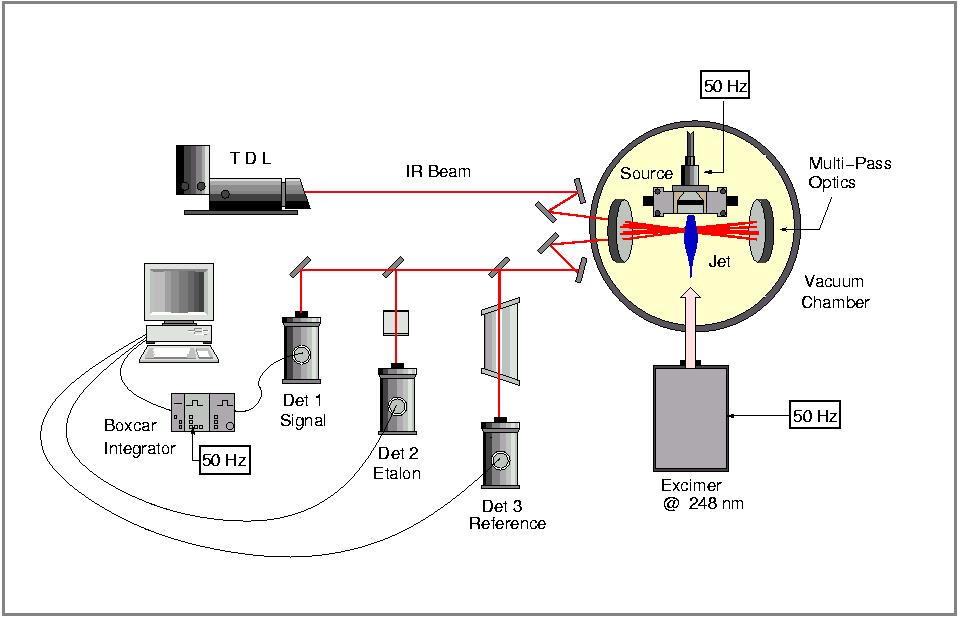
The Carbon Cluster IR spectrometer at Cologne, 2002.
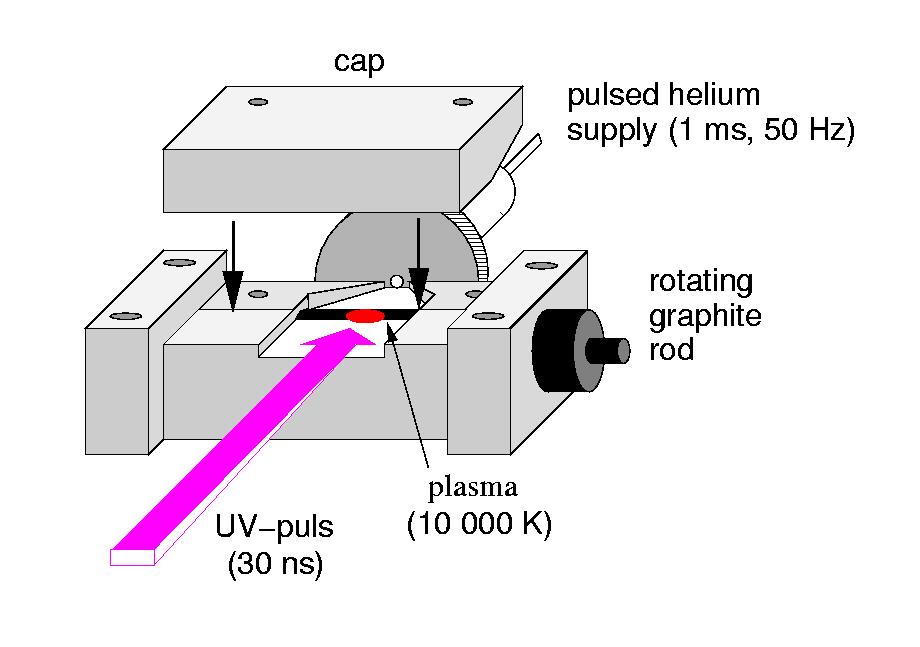
Furthermore investigations on the development of molecular radical sources have been performed. The availability of efficient molecular sources are of great importance for absorption and emission experiments. Hence, their characterization and optimization is indispensable for the success of these kinds of experiments. The excimer laser ablation source used in Cologne is very efficient in the production of pure carbon molecules, i.e. carbon clusters.
|  | |
Scheme of the Cologne Laser Ablation Source, 2000. | Basel Discharge Source during operation, 2002. |
For a further improvement of the production rates new methods were tested. During that work an excimer laser ablation was compared with a Nd:YAG laser ablation using a quadrupole mass spectrometer. Furthermore, investigations on a slit nozzle discharge source have been performed. This type of molecule source is able to produce pure carbon clusters but was originally used for the production of hydro-carbon molecules. Both kinds of molecule sources, i.e. laser ablation as well as slit nozzle discharge sources, produce a plasma which causes significant problems when recording mass spectra. Therefore, a mass spectrometer specially designed for plasma applications in combination with the discharge slit nozzle has been utilized. Cations as well as anions could be detected but no signal of discharge related neutrals were found so far.
Acknowledgements
Many tanks to T. Giesen, G. Winnewisser, U. Fuchs for their support and motivation.
Far-IR studies of molecular ion clusters
My research project at Berkeley was to study molecular ion clusters, for example, protonated water clusters like H+(H2O)n. Because of the production method (discharge of water vapor) an isolated study of these species is very difficult and nearly always requires precise knowledge of the neutral water clusters or other species created during this process. The development of an experimental setup combining an far infrared laser spectrometer, a discharge supersonic jet and velocity modulation technique was extremely challenging but would ultimately be an effective tool to distinguish between neutral and ionic species.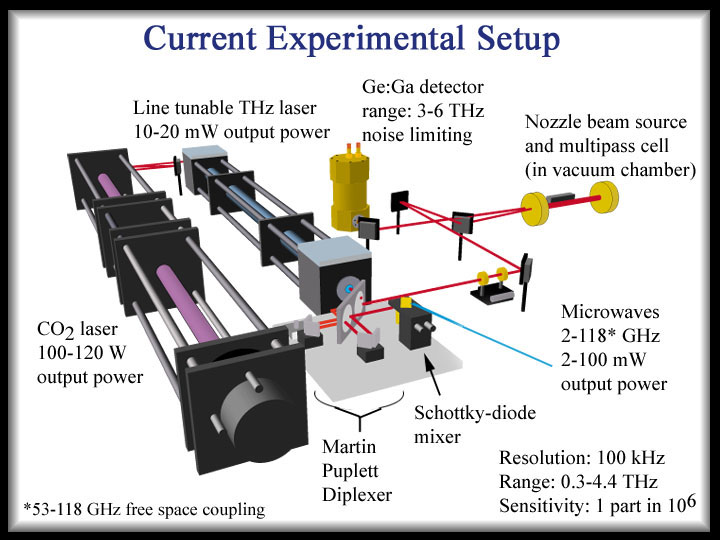
(The Berkeley FIR-laser spectrometer, 2003)
Acknowledgements
This work was supported by the Alexander von Humboldt Foundation via the Feodor Lynnen program.
Research related activities
| 05/2005 | Cooperation with FZ Juelich concerning a hydrogen atom source |
| 03/2004 | Guest stay at the BIMA telescopes, Hat Creek California |
| 03/2003 | Observations at the 15m SEST telescope in La Silla, Chile |
| 09/2002, 07/2003 | Observations at the 30m IRAM telescope, Pico Veleta, Sierra Nevada, Spain |
| 11/2000 | Research stay at Balzers/Lichtenstein, analysis of discharge plasmas |
| 11/2000 | Guest stay at the Institute for Chemical Physics, University Basel, Prof. J. Maier |
| 10/1996 | Guest stay at the large optical telescopes in Sutherland (SAAO), South Africa |
 Harvard-Smithsonian Institution
Harvard-Smithsonian Institution

 Nederlandse Organisatie voor Wetenschappelijk Onderzoek
Nederlandse Organisatie voor Wetenschappelijk Onderzoek
 Nederlandse Onderzoekschool Voor Astronomie
Nederlandse Onderzoekschool Voor Astronomie
